Editas Medicine, a biotechnology company, has received a “Buy” rating from 19 research firms. Of those, three rated the stock as “Sell,” seven as “Buy” and four as “Buy.” The average price target for the next 12 months is $15.33. Recent reports have lowered Editas Medicine’s price target from $36.00 to $30.00 and raised it from $13.00 to $15.00. Editas Medicine’s stock opened at $6.71 and has a 50-day moving average of $8.70 and a 200-day moving average of $9.86. The company’s market capitalization is $462.79 million, and it has a PE ratio of -2.09 and a beta of 1.86. Institutional investors and hedge funds have recently added or reduced their positions in Editas Medicine.
In its latest quarterly earnings report, Editas Medicine posted earnings per share of ($0.88), missing the consensus estimate by ($0.04). The company had a negative net margin of 1,118.32% and a negative return on equity of 50.28%. Editas Medicine’s revenue for the quarter was $6.54 million, compared to the consensus estimate of $3.76 million. Over the last year, the company’s revenue has declined by 47.6%. Analysts predict that Editas Medicine will post earnings per share of -3 for the current year.
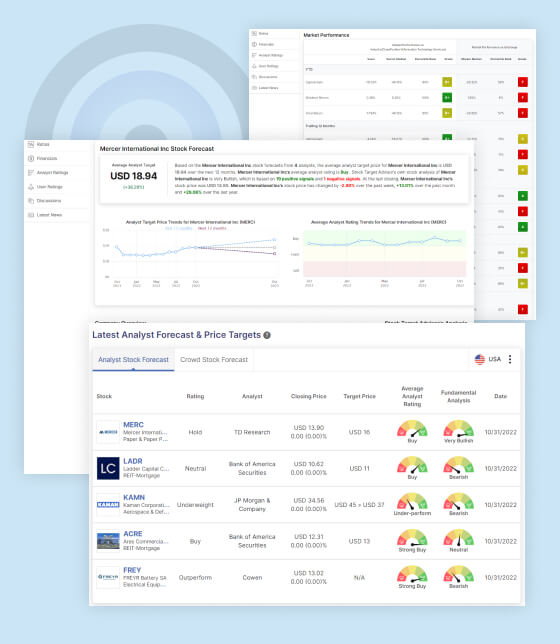
Editas Medicine Stock Forecast
According to 19 analysts’ Editas Medicine Inc stock forecast, the average target price for the next 12 months is $13.88, with an average analyst rating of Buy. However, Stock Target Advisor’s own analysis of Editas Medicine Inc is Bearish, based on 1 positive signal and 3 negative signals. As of the last closing, Editas Medicine Inc’s stock price was $6.71, representing a -3.73% change over the past week, -14.30% change over the past month, and a -61.81% change over the past year.
Company Overview
Editas Medicine, Inc. is a clinical stage biotechnology company that is focused on developing genomic medicines to treat various severe diseases. The company is known for its CRISPR-based gene editing platform, which is used to develop its proprietary medicines. Editas Medicine is currently developing EDIT-101, which is in Phase 1/2 BRILLIANCE trial for Leber Congenital Amaurosis 10, and EDIT-301, a gene-edited medicine for sickle cell disease and transfusion-dependent beta-thalassemia and is based in Cambridge, Massachusetts, United States.